Nature of Thermal Spray Coatings
Contents:
Nature of Thermal Spray Coatings
What is a thermal ( flame ) spray coating? A coating produced by a
process in which molten or softened particles are applied by impact
onto a substrate.
A common feature of all thermal spray coatings is their lenticular
or lamellar grain structure resulting from the rapid solidification of
small globules, flattened from striking a cold surface at high
velocities.
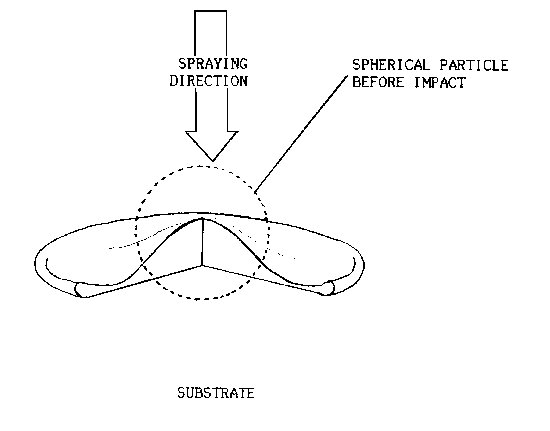
Fig.l Schematic diagram of thermally sprayed spherical particle
impinged onto a flat substrate
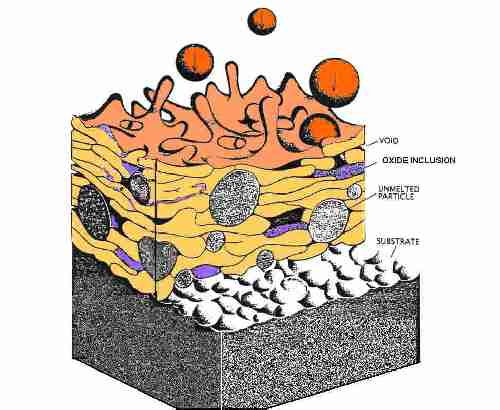
Schematic Diagram of Thermal Spray Metal Coating
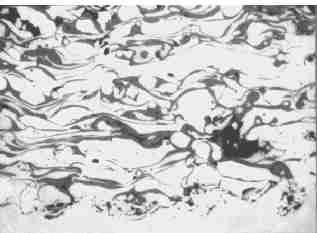
Fig.2. A typical microstructure of a metallic thermally sprayed coating. The lamellar structure is interspersed with oxide inclusions and porosity.
Links to other Photomicrographs
BONDING.
back to contents)
The bonding mechanisms at the thermal spray coating/substrate
interface and between the particles making up the thermal spray coating
is an area which in many cases is still subject to speculation. It
generally suffices to state that both mechanical interlocking and
diffusion bonding occur.
Thermal Spray Coating Bonding Mechanisms:
- Mechanical keying or interlocking.
- Diffusion bonding or Metallurgical bonding.
- Other adhesive, chemical and physical bonding mechanisms -oxide
films, Van der Waals forces etc..
Factors effecting bonding and subsequent build up of the coating:
- Cleanliness
- Surface area
- Surface topography or profile
- Temperature ( thermal energy )
- Time ( reaction rates & cooling rates etc.. )
- Velocity ( kinetic energy )
- Physical & chemical properties
- Physical & chemical reactions
Cleaning and grit blasting are important for substrate preparation.
This provides a more chemically and physically active surface needed
for good bonding. The surface area is increased which will increase the
coating bond strength. The rough surface profile will promote
mechanical keying.
Individual particle cooling rates on impact can be of the order of 1
million º C per second (106Ks-l).
Thermal interaction is obviously very limited. Important with regard to
diffusion bonding (temperature and time dependent).
Increase in thermal and kinetic energy increases chances of
metallurgical bonding. (temperature, velocity, enthalpy, mass, density
and specific heat content etc.. ). Thermal spray materials like
Molybdenum, Tungsten, and Aluminium / metal composites produce so
called "self bonding" coatings. These materials have comparatively high
bond strengths (increased metallurgical or diffusion bonding ) and can
bond to clean polished substrates
Molybdenum and other refractory metals have very high melting points
thus the interaction between substrate and coating particles will be
increased due to the higher temperatures involved and longer cooling
cycles. Also molybdenum oxide volatilizes and does not get in the way
of metallurgical bonding.
Aluminium / metal composites produce increased levels of exothermic
reaction due to reactions of aluminium with metals like nickel to
produce nickel aluminide and with oxygen producing aluminium oxide. The
increased thermal action increases degree of diffusion bonding.
Higher preheat temperatures for the substrate increase diffusion
bonding activities but will also increase oxidation of the substrate
which could defeat the objective of higher bond strengths.
High kinetic energy thermal spraying using HEP, HVOF and cold spray
produce high bond strengths due to the energy liberated from high
velocity impacts. The high density tungsten carbide/cobalt and cold
spray coatings are good examples.
Metallurgical or diffusion bonding occurs on a limited scale and to
a very limited thickness (0.5 µm max. with heat effected zone @
25µm) with the above type coatings.
Fused coatings are different. These are remelted and completely
metallurgically bonded with the substrate and its self.
COATING STRUCTURE
(back to contents)
High cooling rates or super cooling (106 Ks-l)
of particles can cause the formation of unusual amorphous (glassy
metals) microcrystalline and metastable phases not normally found in
wrought or cast materials.
A large proportion of thermal spraying is conducted in air or uses
air for atomisation. Chemical interactions occur during spraying,
notably oxidation. Metallic particles oxidise over their surface
forming an oxide shell. This is evident in the coating microstructure
as oxide inclusions outlining the grain or particle boundaries. Some
materials (such as titanium) interact with or absorb other gases such
as hydrogen and nitrogen.
Coatings show lamellar or flattened grains appearing to flow
parallel to the substrate. The structure is not isotropic, with
physical properties being different parallel to substrate
(longitudinal) than across the coating thickness (transverse). Strength
in the longitudinal direction can be 5 to 10 times that of the
transverse direction.
The coating structure is heterogeneous relative to wrought and cast
materials. This is due to variations in the condition of the individual
particles on impact. It is virtually impossible to ensure that all
particles are the exact same size and achieve the same temperature and
velocity.
All conventionally thermally sprayed coatings contain some porosity
(0.025% to 50% ). Porosity is caused by:
- Low impact energy ( unmelted particles / low velocity )
- Shadowing effects ( unmelted particles / spray angle )
- Shrinkage and stress relieve effects
The above interactions can make the coatings very different from
their starting materials chemically and physically.
STRESS
(back to contents)
Cooling and solidification of most materials is accompanied by
contraction or shrinkage. As particles strike they rapidly cool and
solidify. This generates a tensile stress within the particle and a
compressive stress within the surface of the substrate. As the coating
is built up, so are the tensile stresses in the coating. With a lot of
coatings a thickness will be reached where the tensile stresses will
exceed that of the bond strength or cohesive strength and coating
failure will occur.
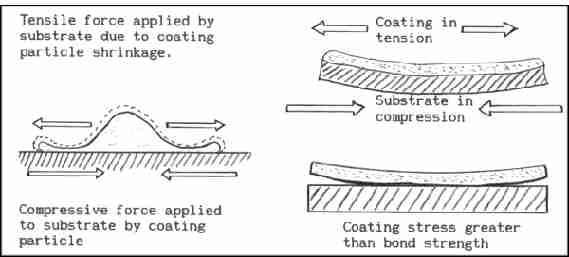
High shrink materials like some austenitic stainless steels are
prone to high levels of stress build up and thus have low thickness
limitations. Look out for thickness limitation information on coating
data sheets. Generally thin coatings are more durable than thick
coatings.
Spraying method and coating microstructure influence the level of
stress build up in coatings. Dense coatings are generally more stressed
than porous coatings. Notice that Combustion powder sprayed coatings
generally have greater thickness limitations than plasma coatings.
Contrary to that just mentioned, the systems using very high kinetic
energy and low thermal energy (HVOF, HEP, cold spray) can produce
relatively stress free coatings that are extremely dense. This is
thought to be due to compressive stresses formed from mechanical
deformation (similar to shot peening) during particle impact
counteracting the tensile shrinkage stresses caused by solidification
and cooling.
PROPERTIES
(back to contents)
Compare properties of unsuported coatings with wrought/cast bulk equivalents:
| PROPERTY | COATING | WROUGHT/
CAST |
| | |
| Strength | low (5-30%) | high (100%) |
| Ductility | very low (1-10%) | high (100%) |
Impact
strength | low | high |
| Porosity | high | low |
| Hardness | higher particle/
micro-hardness | higher bulk/
macro-hardness |
Wear
Resistance | high | low |
Corrosion
Resistance | low | high |
| Machining | poor | good |
This table shows a very general relative trend. There will always be exceptions.
This comparison generally shows coating properties in a bad light,
and does not take into consideration that coatings are usually
supported by a substrate. Coatings are generally only used to give
surface properties such as wear resistance and not to add strength.
Remember, bulk strength supplied by the substrate (cheap, strong and
ductile). Surface properties supplied by the coating ( wear and
corrosion, etc..). Due to the small quantity of material required for a
coating, more exotic materials can be used economically. The properties
of some coatings cannot be fabricated by any other method.
Properties of coatings should be considered in their own right and
not the properties of the original material prior to spraying as they
can be very different physically and chemically.
Porosity
(back to
contents)
This is present in most thermally sprayed coatings (except VPS, post
heat treated coatings or fused coatings). 1 to 25% porosity is normal
but can be further manipulated by changes in process and materials.
Porosity can be detrimental in coatings with respect to:
- Corrosion - (sealing of coatings advised).
- Machined finish.
- Strength, macrohardness and wear characteristics.
Porosity can be important with respect to:
- Lubrication - porosity acts as reservoir for lubricants.
- Increasing thermal barrier properties.
- Reducing stress levels and increasing thickness limitations.
- Increasing shock resisting properties.
- Abradability in clearance control coatings.
- Applications in prosthetic devices and nucleate boiling etc..
Oxide
(back to contents)
Most metallic coatings suffer oxidation during normal thermal
spraying in air. The products of oxidation are usually included in the
coating. Oxides are generally much harder than the parent metal.
Coatings of high oxide content are usually harder and more wear
resistant. Oxides in coatings can be detrimental towards corrosion,
strength and machinability properties.
Surface Texture
(back
to contents)
Generally the as-sprayed surface is rough and textured. The rough
and high bond strength coatings are ideal for bond coats for less
strongly bonding coatings. Many coatings have high friction surfaces
as-sprayed and this property is made use of in many applications
(rolling road drum surfaces for MOT brake testing). Some plasma sprayed
ceramic coatings produce smooth but textured coatings important in the
textile industry. Other applications make use of the abrasive nature of
some coating surfaces. Thermally sprayed coatings do not provide bright
high finish coatings with out finishing like that of electroplated
deposits.
Strength
(back to contents)
Coatings generally have poor strength, ductility and impact
properties. These properties tend to be dictated by the "weakest link
in the chain" which in coatings tends to be the particle or grain
boundaries and coating/substrate interface. Coatings are limited to the
load they can carry, and thus require a substrate for support, even
then, coatings are poor when point loaded.
Internal tensile coating stresses generally adversely effect
properties. Effective bond strength is reduced and can be destroyed by
increasing levels of internal stress. This in turn effects coating
thickness limits. Coatings on external diameters can be built up to
greater thickness than that on internal diameters.
Surface properties such as wear resistance are usually good, but the
properties are more specific to the material or materials used in the
coating. The properties of a substrate need only to be strength, ease
of fabrication and economic (like mild steel). The coating supplies the
specific surface properties desired. For example, materials used for
applications of thermal barrier and abradable clearance control by
nature have poor strength and thus benefit from being applied as a
coating onto a substrate which supplies the strength.
Some Properties Thermally Sprayed Coatings can Provide:
- Tribological (wear, resistance).
- Corrosion resistance.
- Heat resistance.
- Thermal barrier.
- Electrical conductivity or resistivity
- Abradable or abrasive.
- Textured surfaces.
- Catalyst and prosthetic properties,
- Restoration of dimension.
- Copying of intricate surfaces.
(back to contents)
NON-DESTRUCTIVE TESTING OF
THERMALLY SPRAYED COATINGS.
There are very few reliable NDT methods available for thermally
sprayed coatings. The majority of tests for coatings tends to be of a
destructive nature, which, obviously can not be used on the actual
coated part going into service and therefore, must be considered as a
test for process control.
The main practical NDT methods used are:
- Dimensional measurements- micrometer, eddy current and magnetic
thickness measuring devices etc..
- Machining tests-response of coating during machining operations
is a good test for general integrity.
- Visual inspection- grit blast, spraying, coating/substrate,
machined finish.
- Dye penetrant- used in limited applications, but natural coating
porosity fogs flaw indications.
Ultrasonic and magnetic particle flaw detection methods have proved
to be poor with thermally sprayed coatings due to the very high number
of particle boundaries giving flaw like responses and causing high
levels of interference.
Hardness testing is generally considered a destructive test for
coatings unless made in a non-working area.
Advanced techniques like thermography, Thermal wave interferometry
and acoustic emission are presently being researched and are still
laboratory set-ups with limited practical use for industry.
Destructive testing such as hardness, bend, bond strength,
metallography etc.. are important to prove the process and coating
integrity expected in the component.
The limited non-destructive testing available for thermally sprayed
coatings should emphasise the need for a high standard of quality
control over the process, to ensure a high level of confidence in the
coated products.
(back to
contents)
Factors Effecting The Thermal Spray
Coating Process

(back
to contents)


Telephone: +44 (0)1252 405186
Email: tsc@gordonengland.co.uk
Site Links
Introducing
Nature of Thermal Spray Coatings
Surface Engineering in a Nutshell
Surface Engineering Forum
Thermal Spray Gun Repair Service
Plasma Consumable Parts
Thermal Spray Powder Supplies
Applications:
Thermal Spray Coatings on Carbon and Glass Fibre Reinforced Polymers
HVOF Coating of Paper Making Roll
Abradable Coatings
Photomicrographs
Thermal Spray Processes:
Combustion Wire Thermal Spray Process
Combustion Powder Thermal Spray Process
Arc Wire Thermal Spray Process
Plasma Thermal Spray Process
HVOF Thermal Spray Process
HVAF Thermal Spray Process
Detonation Thermal Spray Process
Plasma Flame Theory
Cold Spray Coating Process
Wear and Use of Thermal Spray Coatings
Corrosion and Use of Thermal Spray Coatings
Glossary of Thermal Spray and Surface Engineering Terms
Image Directory for Thermal Spray Coatings
Plasma Gas Flow Information
Plasma Gas Flow Correction Calculator
Contact Form
Links to other interesting sites related to thermal spray and surface engineering
Reciprocal Links
Periodic Table of the Elements
SI Units
Calculators for Conversion between Units of Measurement
Hardness Testing
Surface Engineering Message Board Archive
Surface Engineering Message Board Archive Index
Photography Gallery
Photography Gallery3
© Copyright Gordon England






