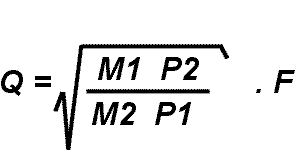
Formula for Plasma Gas Flow Conversion



| Molecular Weights of Gases: | Air | 29 |
| Nitrogen | 28 | |
| Argon | 40 | |
| Helium | 4 | |
| Hydrogen | 2 |
| Primary gas flowmeters | Nitrogen @ 50 psi, 70oF |
| Secondary hydrogen flowmeters | Hydrogen @ 50 psi, 70oF |
| Secondary helium flowmeters | Nitrogen @ 50 psi, 70oF |